

 |
 |
Invertebrates
| A note on butterflies utilising longan fruit | |
| New records of epilithic cyanobacteria: a furthur documentation of rocky shore species richness |
New records of epilithic cyanobacteria: a further documentation of rocky shore species richness
by Sanjay Nagarkar
As we approach November, the algal assemblages on rocky shores seem to be more comfortable after facing harsh physical environmental conditions during the summer (June to September). Many of these were cyanobacteria, well able to tolerate these harsh conditions and that served as the only primary producers on rocky shores. The situation in winter, however, when environmental conditions are favourable for algal growth is very different. At this time of the year (November-March), Hong Kong rocky shores are densely populated by a variety of micro- and macroalgal species, of which the proportion of microalgal species is much higher. These species comprise cyanobacteria, diatoms and the spores and sporelings of macroalgae, that inhabit the entire tidal gradient in the form of an epilithic biofilm, exhibiting distinct zonation patterns. In the biofilm, cyanobacteria are the most important primary producers due to their high nutritional quality, fast production rate and great species diversity. A wide salinity gradient, oligotrophic and eutrophic waters and different rock types, i.e., basalt, granodiorite and volcanic tuff on Hong Kong rocky shores, provide a favourable environment to accommodate a large variety of epilithic cyanobacterial species.
In the past four years, I have surveyed the coastline of Hong Kong to document cyanobacteria species richness and biodiversity. So far, I have recorded more than 200 species, many of which are believed to be new to science (Nagarkar 1998, 2000, 2001, 2002). During this survey I made an interesting observation. In winter, at mid-shore levels, the biofilms become thicker and are mostly composed of non-heterocystous, filamentous, cyanobacteria, especially a species of the genera Lyngbya, Oscillatoria and Phormidium (LOP). These species are the major food items for intertidal grazers in the mid-shore. Morphological investigation of the thick LOP biofilm revealed a total of 32 filamentous species from 26 locations around Hong Kongs rocky shores (Fig. 1, 2), all of which are new records for Hong Kong (Table 1). The addition of these species takes the Hong Kong cyanobacteria checklist to 87 published species. At present, I am compiling unicellular and heterocystous cyanobacteria species richness lists. Please dont forget to check your next issue of Porcupine! for an update on Hong Kongs cyanobacteria diversity.
Bibliography
Nagarkar, S. (1998). New records of marine cyanobacteria from rocky shores of Hong Kong. Bot. Mar. 41: 527-542.
Nagarkar, S. (2000). Morphology and ecology of epilithic coccoid marine cyanobacteria from Hong Kong. Asian Mar. Biol 17: 15-24.
Nagarkar, S. (2001). New records of intertidal epilithic cyanobacteria from Hong Kong rocky shores. Algae 16(4): 343-348.
Nagarkar, S. (2002). Morphology and ecology of new records of cyanobacteria belonging to the genus Oscillatoria from Hong Kong rocky shores. Bot. Mar. 45: 274-283.
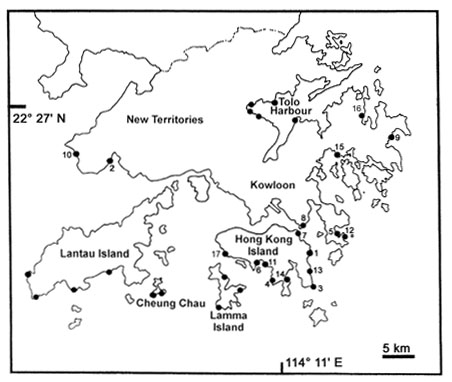 |
| Fig. 1. Location of rocky shores survey areas around Hong Kong (· sampling site). Principal sites are numbered as follows: 1. Big Wave Way, 2. Butterfly Beach, 3. Cape dAguilar, 4. Chung Hum Kok, 5. Clear Water Bay, 6. Deep Water Bay, 7. Heng Fa Chuen, 8. Lei Yue Mun, 9. Long Ke Wan, 10. Lung Kwu Tan, 11. Middle Bay, 12. Shek Mei Tau, 13. Shek O, 14. tai Tam, 15. Stanley Bay, 16. Tai Mong Tsai, 17. Tai Tan, 18. Wah Fu. |
Table 1. Check list of epilithic cyanobacteria; genera Lyngbya, Oscillatoria and Phormidium.
| Lyngbya cf. aeruginosa-coerulea Kützing ex Gomont, |
| Lyngbya cf. aesturaii var. constricta Ghose, |
| Lyngbya cf. birgei Smith G. M. |
| Lyngbya cf. cinerescens Kützing |
| Lyngbya cf. hieronymusii Lemm. |
| Lyngbya cf. major Meneghini ex Gomont |
| Lyngbya cf. martensiana var. minor Gardner |
| Lyngbya cf. stagnina Kützing |
| Lyngbya cf. truncicola Ghose |
| Oscillatoria cf. annae van Goor ex Gomont |
| Oscillatoria bonnemaisonii Crouan ex Gomont |
| Oscillatoria cf. borneti (Zukal) Forti |
| Oscillatoria brevis Kützing ex Gomont |
| Oscillatoria chalybea Mertens in Jürgens ex Gomont |
| Oscillatoria cf. curviceps Agardh ex Gomont |
| Oscillatoria cf. irrigua Kützing ex Gomont |
| Oscillatoria cf. limosa Agardh ex Gomont |
| Oscillatoria margaritifera Kützing ex Gomont |
| Oscillatoria cf. obtusa Gardner |
| Oscillatoria cf. ornata Kützing ex Gomont |
| Oscillatoria cf. princeps Vaucher ex Gomont |
| Oscillatoria cf. proboscidea Gomont |
| Oscillatoria cf. salina Biswas |
| Oscillatoria sancta Kützing ex Gomont |
| Phormidium ambiguum Gomont |
| Phormidium anomala Rao C. B. |
| Phormidium stagnina Rao, C. B. |
| Phormidium retzii (Ag.) Gomont |
| Phormidium lucidum Kütz. ex Gomont |
| Microcoleus acutissimus Gardner |
| Microcoleus chthonoplastes Thuret ex Gomont |
| Hydrocoleum lyngbyaceum Kütz. ex Gomont |
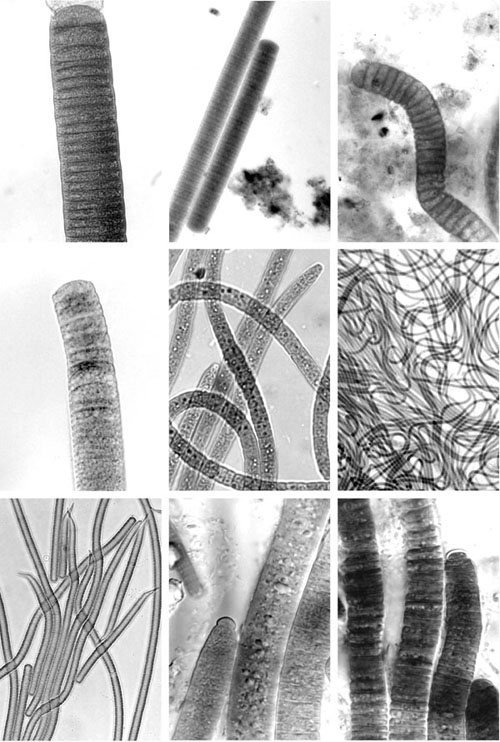 |
|
Fig. 2. Some examples of epilithic marine cyanobacteria (LOP); size (width) ranges from 10- 50 µm. |
|
|
P.7-8 |
|
Porcupine! |
 Copyright © 2000 |